CDC and Missouri Report Recovered Human H5 Case with No Known Infected Animal Contact
Sequencing needed for more analysis; other studies show widespread H5N1 2.3.4.4b clade adaptation in mammals, widening list of potential viral sources
Friday evening CDC reported the following: CDC Confirms Human H5 Bird Flu Case in Missouri | CDC Newsroom
Separately, the Missouri Department of Health and Senior services released the following statement: News Releases | Health & Senior Services (mo.gov)
As both pieces stated the investigations are in very early stages, however, the following facts are both known and reassuring:
Missouri DHSS reports that the patient, who was hospitalized, had underlying medical conditions, was treated with influenza antiviral medications, subsequently discharged, and has recovered.
There is no known animal exposure associated with the infection at this time.
No ongoing transmission among close contacts or within the area has been identified to date.
While this is the 14th human case of H5 reported in the United States during 2024, it is the first case of H5 without known occupational exposure to sick or infected animals. To be clear, at this point CDC has not stated that this is H5N1 (the neuraminidase has not been subtyped) and no details have been shared regarding the H5 clade or avian pathogenicity class, pending further sequencing work. So, it is premature to even state that this is an H5N1 infection, let alone that it is the H5N1 2.3.4.4b B3.13 (dairy) clade. Hopefully CDC will be able to successfully sequence and grow the isolate in a few days for further work.
If this human isolate is the recent prominent B3.13 clade that is spreading so rapidly in multiple species of wildlife, dairy herds and poultry flocks, then this independent human isolation a major development that raises multiple questions for CDC:
Is H5N1 already circulating in humans at some very low level despite little evidence for that in existing influenza surveillance systems prior to this case?
Are there unexplained animal reservoirs shedding H5N1 into the environment at levels sufficient to infect humans, especially those with underlying medical conditions?
Alternatively, if the isolate is closely related to current dairy isolates and if the patient consumed dairy products, could there be failures in viral inactivation in consumer milk products?
Are there further structural changes in the isolated virus making human infection more likely?
Further progress depends upon characterizing the viral isolate from the patient and seeking the patient’s cooperation in understanding and testing their environment and living surroundings for possible sources of infection. Much of this must happen outside of public view due to confidentiality provisions in public health investigations.
Regardless, CDC will disclose the genetic characteristics of the viral isolate, if they can obtain it. That information will also be of great value to the livestock community in assessing the degree to which the H5 2.3.4.4b B3.13 “dairy virus” may or may not be involved in this case. Until we have those pieces of information, further speculation regarding dairy cattle or milk’s role in this case are extremely premature. Even if it IS that viral strain, it’s rapid spread into multiple species makes zoonotic source attribution for human infection increasingly challenging.
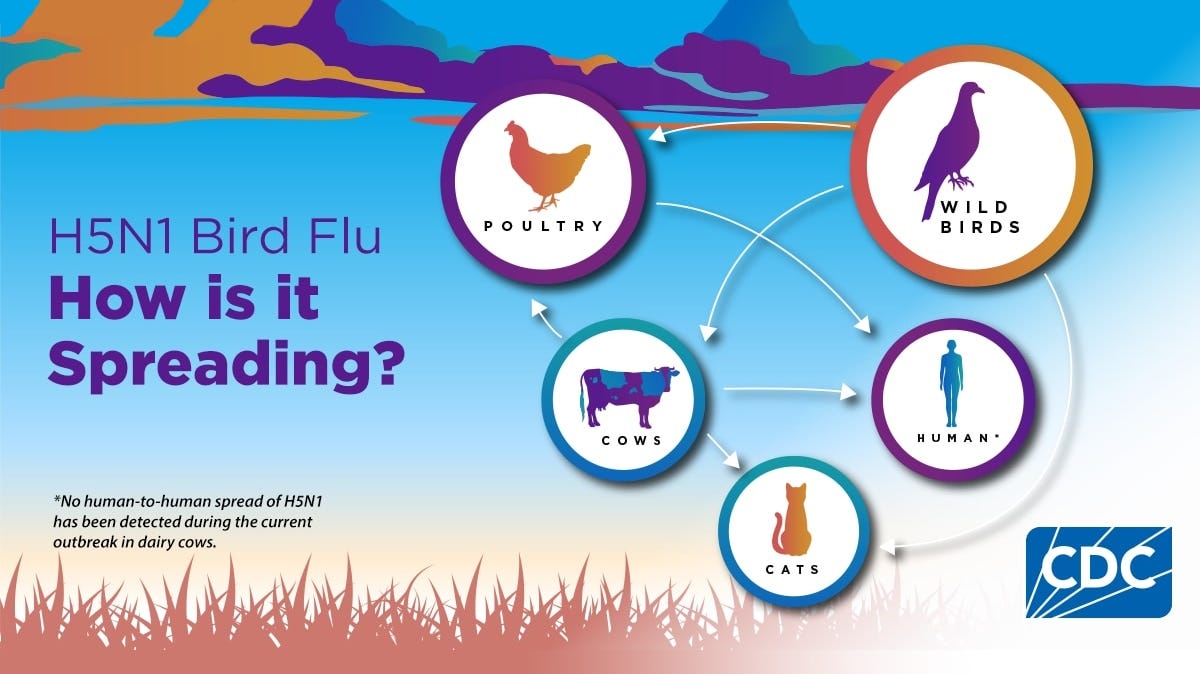
The Washington Post Friday published an extended H5 Q and A piece: Bird flu explained: How it spreads, milk and egg safety and more. This is a well-informed piece related to H5 and our agricultural industries. Traditionally, issues covered have been the extent of our concerns. Even the current CDC graphic shown here shows potential human infection being limited to contact with poultry or cows:
However, we’ve had several articles and research releases in the past few weeks related to other species susceptibility to and carriage of H5N1. Michael Coston published the following blog this past week reviewing several papers:
Some of these references run a bit far afield from this week’s topic of potential source(s) for a human H5N1 infection; however, we may need to be a bit more “open-minded” when we are considering potential routes for unexplained human infections in the face of burgeoning environmental sources of virus.
Thinking back to Colorado in late summer, we had a mini H5 cat mortality epidemic in the same time period as dairy and poultry H5 outbreaks. Simultaneously multiple H5-positive rodents were collected, which can carry virus, and which cats may “harvest”. While the cattle and poultry amplified large volumes of virus into the environment, that amplification was likely reflected in the surrounding peri-domestic population in a wider area for an unknown period of time. The net effect may be additional sources of risk for mammalian H5N1 spread. I’m not aware that we yet have good estimates related to the range of peri-domestic infection, nor its duration following viral introduction into an area’s population.
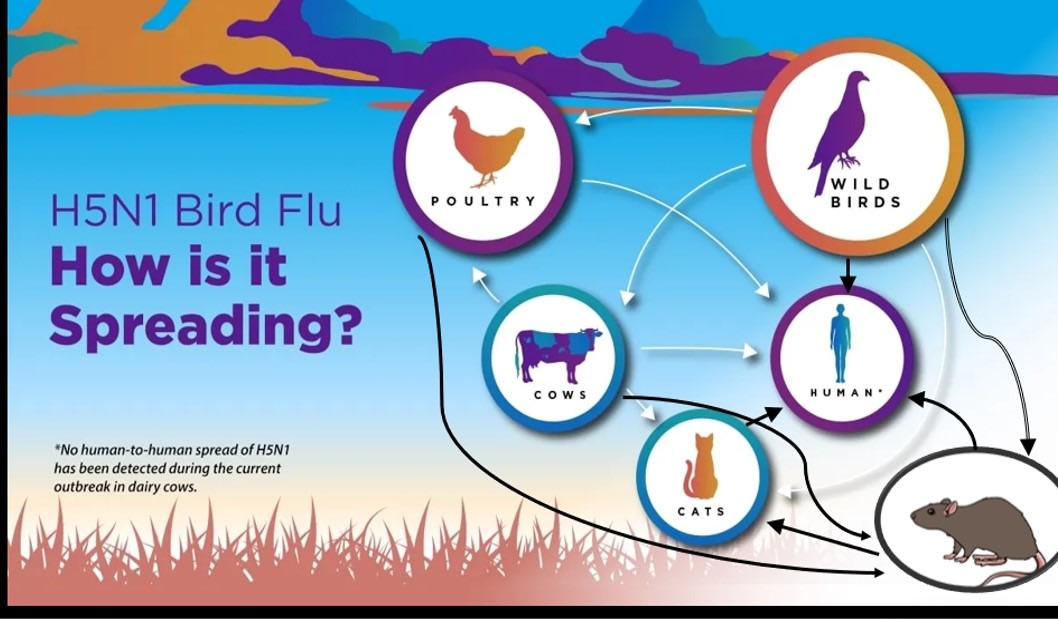
So back to the CDC graphic above, we need to add several peri-domestic species to the mix, including rodents who live much more intimately with humans than we’d like to admit. The arrows for transmission to humans likely need to extend both from cats and from peri-domestic mammals, as well as from some birds to humans on an occasional basis:
Possible Additional Routes of Human Exposure through rodents/peri-domestic spp.:
I’m not trying to co-opt the CDC’s messaging. I believe the intent of the graphic is likely to illustrate known or documented routes of spread to humans. My added arrows are much more speculative; however, it’s still helpful to think outside of the known when the cases no longer fit.
As a final thought to this week’s column, I’m not sure we’ve fully come to terms with the significance of the risk that “mammalization” of H5N1 2.3.4.4b has brought to our livestock industries. This is not a stable virus, it is a potential zoonotic virus, and it has happily found a home in a series of species which have never dealt seriously with influenza in the past. Symptoms seem to range from non-clinical to mastitis, to encephalitic death, depending on the species and viral sequences and reassortments. We already know how devastating it has been for some marine mammal species and their ecosystems.
As it settles into our domestic species, and into some peri-domestic birds and mammals that surround us, I have to wonder how we’ll manage the crossover risks. Nature always finds a balance eventually; that will happen with H5N1. However, we have multiple U.S. livestock production systems designed to exploit economies of scale with limited biosecurity processes for controlling bacterial and stable viral pathogens but are horribly susceptible to shifting RNA viral pathogens like influenza. With an apparent rapidly growing list of reservoirs for H5N1, how will we manage our animals’ risk for this agent?
John