H5N1 in Cattle at 3 months-where are we?
Critical information is missing in hopes that infected herd "virus exhaustion" and enhanced biosecurity will eliminate H5N1 in cattle, all evidence to the contrary notwithstanding.
Science published an article this past week reviewing the state of the battle against H5N1 in the U.S. Dairy Industry:
Is ‘cow flu’ here to stay? Three months after it emerged, fears are growing | Science | AAAS
“More than 3 months after the first reported H5N1 avian influenza outbreak at a U.S. dairy farm, some researchers are starting to wonder whether the virus is here to stay.
The U.S. government says it, with the help of the dairy industry, is working diligently to prevent that outcome. “We believe if we can stop the movements [of infected cattle], improve the biosecurity, and then help the producers, … we can eliminate this virus,” Rosemary Sifford, chief veterinary officer of the U.S. Department of Agriculture (USDA) said at a 25 June webinar organized by the National Academies of Science, Engineering, and Medicine.
But given the lack of cooperation from the industry and what many see as a lackluster government response, other scientists are doubtful. Sifford’s upbeat outlook “really surprised” veterinarian Michelle Kromm, another presenter at the webinar. “None of what they've done publicly from a policy standpoint would indicate that they're trying to eliminate this,” says Kromm, who was the top turkey vet at Hormel Foods during a devastating H5N1 outbreak in poultry a decade ago. “To me, all stars are aligning to say we've accepted that this is endemic.” …”
At 3 months into the USDA response to this latest mammalian jump of H5N1 2.3.4.4b into domestic cattle, I agree wholeheartedly with Dr. Kromm. Let’s spend a few paragraphs reviewing the many basic steps that have NOT happened since April 1st that would have put us so much further down the path in understanding our options with this virus.
We STILL have no good evidence of the scope of the outbreak in dairy, or evidence of its status in non-lactating domestic cattle. USDA still has not validated an ELISA serological assay for use and seems to be in no hurry to do so. Serology would be useful in estimating prevalence. Some researchers and practitioners likely have some unapproved serological testing results in hand. What is the confidence level in the antibody tests among researchers and practitioners that have used them? Are there legal, market, or funding barriers to obtaining serology assays for more widespread use in cattle? Can existing work be summarized outside of USDA channels to begin a knowledge base for reference over time? Ideally a national prevalence study would be undertaken, or a series of targeted studies done to answer specific questions.
As a related question, is there any scientific reason that all cattle are not susceptible to this virus? Any studies to show that non-lactating beef cattle in close contact with infected dairy herds remain negative to infection and are not potential sources of infection to nearby dairy and poultry operations? Conversely, while beef animals may well show minimal clinical illness if infected, they could still potentially serve as sources of new infections in dairy or poultry herds, or as zoonotic threats in their own right.
We could make much more informed hypotheses regarding these questions if interim or final results were reported from USDA-ARS-NADC and Canadian non-lactating dairy infection/transmission studies; both projects should at least provide progress reports with interim findings:
Is virus shed from respiratory tract, urine, and/or in feces – length of time and viral levels?
What is the likely route of infection and infectious dose (assuming all infection is not intramammary)- oronasal (conjunctival) and/or other? Is aerosolization required for infection
What is the pathogenesis of the infection process in cattle (both lactating and nonlactating)
What is the preclinical shedding time and length of shedding of virus? In milk, nasal, oral, urine, feces, semen (anecdotally PCR+ bulk milk has been reported as much as 10 days prior to clinical presentation of mastitis in affected herds)
What is the degree and ease of ongoing transmission to contact animals, if any -apparent route and role of infectious dose predicting onward transmission
What percentage of non-clinical or subclinical illness is seen in infection studies in both primary infection and contact animals?
What is the estimated time to development of antibody titers, including protective antibodies post infection? Does severity of infection correlate with antibody production and protection?
At this point we need some data on recovered herds. What is the range of resolution of acute infection in affected herds at 90 days out? Animal serological titers? Remaining percentage of animals PCR positive, if any, indicating chronic infection or reinfection? If positive, what are the genomic changes in persistent infections? We can’t assume the virus “exhausts itself” in infected herds without follow-up investigations!
Finally, let’s return to “transparency” for a moment. Early on USDA promised maximum transparency and prompt submission of complete sequences and metadata to GISAID as soon as practical. A quick look recently indicated that GISAID has resorted to using NCBI data w/o most metadata in order to at least build the H5N1 Influenza gene phylogenetic trees as best can be done since early in the outbreak. World zoonotic transparency continues to backslide. Even the U.S. is now unwilling to meet GISAID obligations for open and transparent phylogenetic submissions. No national veterinary authority can retain world respect when it cannot enforce data transparency and reporting within its own borders.
Issues For CDC and FDA to Consider:
Why have we not undertaken a national bulk milk testing scheme or alternatively, an organized retail milk screening program to estimate prevalence and allow some sort of nationalized sequencing process? Is there an issue with federal authority for sequencing samples when no individual producers would be identified? FDA has access to samples at the bottling plant level, and a group in Canada has initiated a similar program without issues.
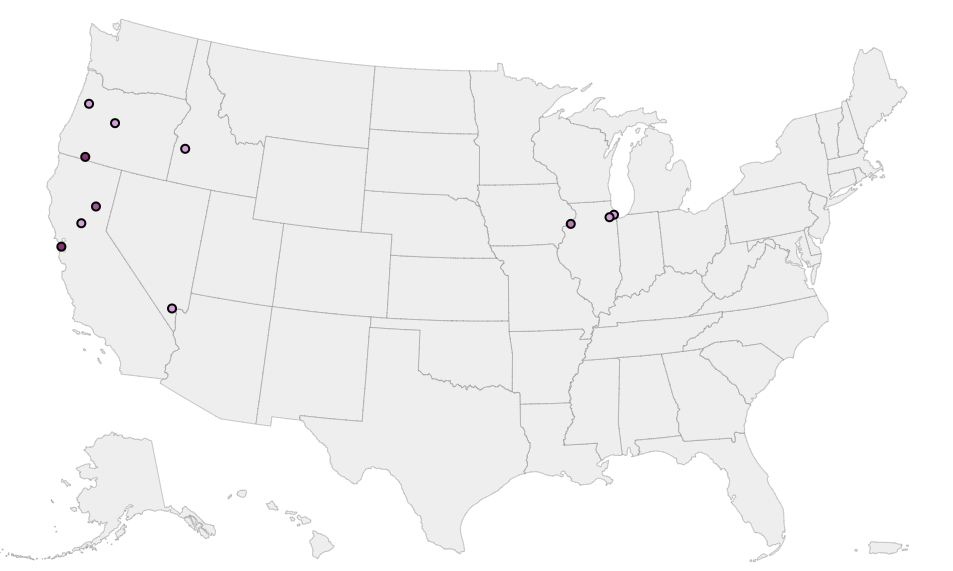
CDC wastewater screening is not being followed up – why are positive summertime matrix samples not sequenced or reported? Here is the latest map from the CDC site with the 2-week maximum levels (June 16-29) for sites with “high moderate” and higher readings plotted on the map (with all other sites hidden for clarity). Last week I called out Illinois and California officials for not following up on sequencing these signals to gain further information. It looks like Oregon, Idaho (in the midst of a dairy outbreak) and Nevada should be added to the list.
The U.S. and world scientific communities remain almost completely at the mercy of dairy industry and USDA voluntary reporting in assessing U.S. H5N1 2.3.4.4b B3.13 evolution. A few poultry outbreak samples may be sequenced as well as assorted wildlife samples, but contemporary cattle sequences found in the current milk supply and wastewater are completely shut off from public analysis and reporting due to an indefensible failure by public health authorities to utilize proven methodologies to generate ongoing contemporary sequences for public health and pandemic preparedness.
It may well be that we were preordained to deal with H5N1 as an endemic disease in cattle; if so, we have wasted a good deal of time and money attempting to set “eradication” policy and procedures that are doomed to fail based on incomplete information and wishful thinking. This virus is here to stay, probably in the entire cattle industry, maybe in all ruminants, and is possibly a risk to the pork industry. We know it is an existential risk to the poultry industry, where area spread from cattle operations has already been devastating to commercial flocks located in proximity to infected dairy herds.
How will we resolve the interspecies pressures between large commercial dairy and poultry entities that this virus has brought to bear? That is the ultimate test for USDA and all of commercial ag, where the competing health and economic storm clouds are gathering.
For all of our animal health issues, please remember we’re also dealing with a rapidly evolving potentially zoonotic virus. If this virus forward transmits even a little bit in people, then we’ll ALL have real problems! Avoiding action in service to economic concerns will not be a great legacy for the ag community.
An article in STAT this morning references ongoing work in multiple labs investigating receptor cell attachment and onward transmission in multiple species:
Debate: Is bird flu virus in cows adapted to better infect humans? | STAT (statnews.com)
Two quotes by prominent influenza researcher taken from the article are worth noting:
The work on predicted binding offers new evidence for wider attachment, including to cells lining the human upper respiratory tract but requires further study to understand the underlying factors, Ian Brown, the former virology head at the UK’s Animal and Plant Health Agency who is now a group leader at the Pirbright Institute, said in a statement to reporters. “Overall the study findings are not unexpected but this report provides further science insight to an evolving situation, that emphasizes the need for strong monitoring and surveillance in affected or exposed populations, both animals and humans to track future risk.”
“It’s not zero transmission; there is some transmission but it’s very limited,” (Yoshihiro) Kawaoka said. That should provide some reassurance that the virus has not yet acquired the ability to easily spread through the air. But how long that will stay true, with the virus expanding its footprint — and with it, opportunities to adapt to human biology — is anybody’s guess.
“Continued surveillance is needed,” Kawaoka said. “We need to be concerned.”
In closing this week, we cannot minimize surveillance or rely only on dated information and sequencing to monitor changes in this virus! We are ALL responsible for insisting that our experts have access to the most recent infectious material available for ongoing monitoring. At 90 days into this it’s time to step up some accountability on all fronts, including our own expectations for our leaders.
John